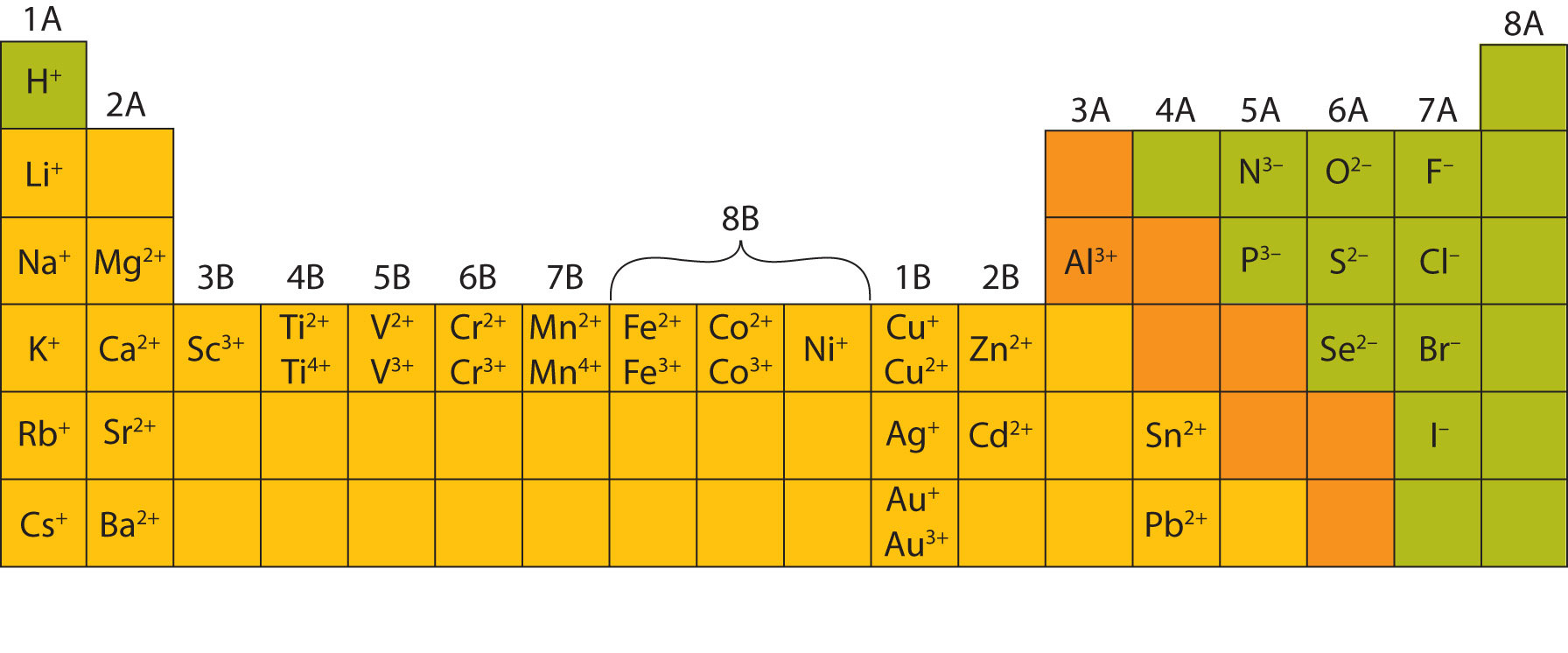
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals. The central atom in our structure will be carbon (it is to the left of oxygen in the periodic table). Consequently, beryllium is significantly smaller than lithium. Oxygen has six valence electrons and carbon has four therefore in CO 3 2 there will be a total of 22 valence electrons, plus two additional electrons from the 2- charge. However, you should be able to determine the ionic state of atoms from the other groups using a Periodic Table. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). charge), giving the ion a net charge of -2. This table also contains the element number, element symbol, element name, and atomic weights of each element. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. This printable periodic table contains the atomic number, element symbol, element name, atomic weights and most common valence charges. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. We will need two hydroxide ions to exactly cancel the 2+ charge on Ca 2 +. (b) The metallic atomic radius, rmet, is. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. From our table, we know that hydroxide has the formula OH and that it carries a 1- charge. Atomic radii are often measured in angstroms (), a non-SI unit: 1 1 × 1010 m 100 pm. Moving from the far left to the right on the periodic table, atoms of main-group metals lose enough electrons to leave them with the same number of electrons as. The Ion Names, Formulas and Charges Chart for chemistry classrooms comprehensively lists the names and formulas of cations and anions, allowing students to. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. Calcium is an alkaline earth metalGroup 2 on the periodic tableso it forms ions with a 2+ charge. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements.
1 Comment
|
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
